Engineered to be Better.
Discovery of the best human therapeutic antibodies starts with AlivaMab Mouse.
Ablexis’ AlivaMab Mouse is the only transgenic animal designed to enable and optimize the efficient discovery and development of the next generation of human therapeutic antibodies.
The platform has been validated for antibody drug discovery by Ablexis and our partners for a range of applications including:
- Challenging targets such as GPCRs
- Challenging design goals such as very low picomolar IC50 values
- Sequence diverse panels of mAbs
- Epitope diverse panels of mAbs
- AlivaMabs being developed in various formats including regular antibodies, bispecifics and CAR-Ts
The AlivaMab Difference
A Better Design Delivers Unique Benefits
Key Features and Benefits of the AlivaMab Mouse
Features
- First and only synthetic, autonomously functioning heavy, kappa and lambda immunoglobulin transgenes
- Unique combinations of human and mouse coding and non-coding elements
- Diverse, curated repertoire of VH, Vκ and Vλ gene segments selected for developability and lowered risk of immunogenicity
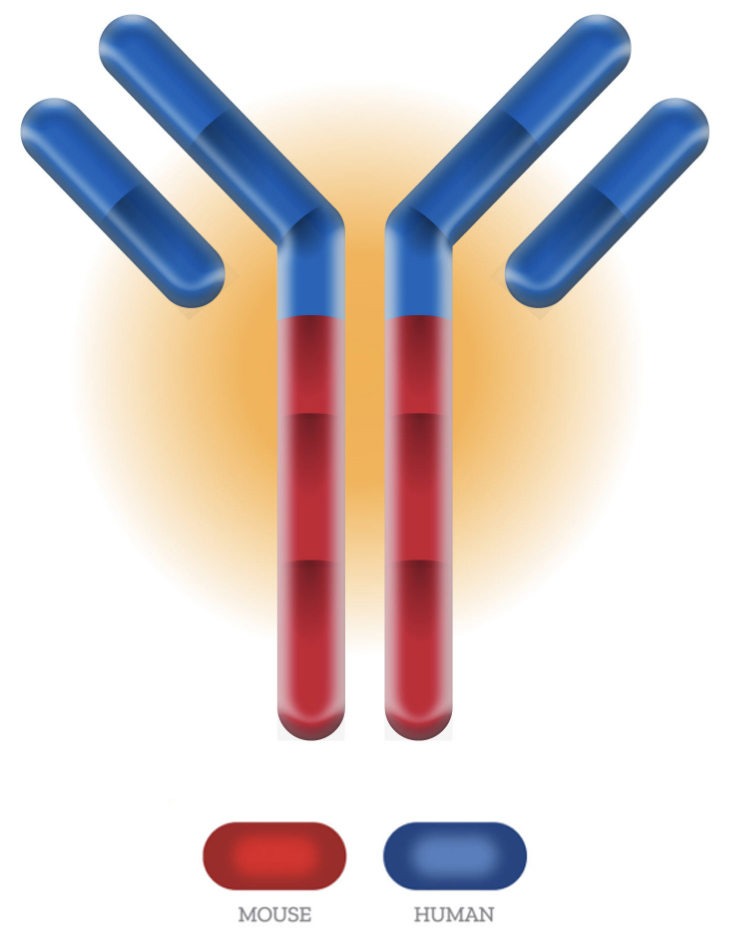
- Patented human/mouse chimeric antibody composition
- Three AlivaMab Mouse strains: K-L with a Igκ/Igλ expression ratio as in humans, Kappa, and Lambda
Benefits
- Robust immune responses for efficient discovery of high-quality therapeutic lead candidates that meet potency, specificity, and developability requirements
- Real diversity in the V region repertoire in both the primary and secondary immune responses better than other transgenic platforms
- Retention of activity and developability qualities upon conversion to fully human
- Choices for AlivaMab Mouse strains allow for maximizing light chain diversity and pre-selecting for light chain composition
AlivaMab Mouse
The superior platform for the discovery of therapeutic antibodies
Your Choice of Platform Matters
The past four decades have seen advances in therapeutic monoclonal antibody discovery technologies from fully mouse to fully human antibodies. Today, fully human antibodies are typically generated using two distinct approaches: (1) in vitro systems and (2) genetically-engineered animals.
However, while humanization and in vitro techniques such as phage, ribosomal, yeast and mammalian display technologies appear simplistically attractive, there is a growing appreciation that downstream risks in development - problems that may not become apparent until after hundreds of millions of dollars have been spent - are amplified by in vitro technologies. Such risks include inadequate potency, CMC issues, off-target binding, and anti-drug antibody formation.
In contrast to in vitro display and humanization technologies, transgenic mice can reduce the risk of expensive problems and failures in later stages of development through harnessing of the natural in vivo immune response to generate high-affinity, high-potency antibodies that inherently possess desirable qualities of developability, specificity, and reduced risk of immunogenicity. This combination of attributes helps explain why there are substantially more marketed fully human antibody drugs that were discovered using transgenic mice than from in vitro display platforms.
Innovative technologies were used to create the specialized transgene designs for the AlivaMab Mouse platform, improving upon the limited bacterial artificial chromosome (BAC) and yeast artificial chromosome (YAC) genomic library approaches of other technologies.
AlivaMab Mouse is the first transgenic animal to use synthetic immunoglobulin (Ig) transgenes, and the only one to use autonomously functioning synthetic Ig transgenes, avoiding the need to hijack the constant regions and cis regulatory elements of the endogenous Ig loci. By first designing the Ig transgenes of AlivaMab Mouse in silico and then synthesizing the DNAs in vitro, multiple aspects were optimized to help meet the challenges of both antibody drug discovery and antibody drug development. These aspects include function of the immunoglobulin transgenes, a thoughtfully curated endowment of VH, Vκ and Vλ gene segments, and a unique part-human/part-mouse composition of the antibody.